Tulip
Frontline operations platform for Life Sciences Manufacturing
Build, adapt, and scale digital operations with a no-code platform — designed for speed, flexibility, and GxP compliance.


Design, deploy, and evolve manufacturing applications with a no-code platform for operational excellence—composable, cloud-native, and highly scalable.
One Platform, Unlimited Use Cases
Build Apps to digitize core processes, data recording, and any forms or supporting process.

Composable Platform
Build flexible, scalable applications that grow with your operations.

No-Code App Editor
Users can create custom, user-friendly applications without coding, turning documents into interactive digital apps.

Guided Workflows
Step-by-step digital work instructions to guide operators, reducing errors and improving efficiency.

Real-Time Data & Analytics
Turn real-time production and machine data into actionable insights that drive smarter decisions.
Integration &
GxP-Ready Ecosystem
A connected, GxP-ready ecosystem that links systems, equipment, and shopfloor data through Tulip.
ERP & eQMS
Trusted data exchange for production and quality
Weighing & Equipment
Various production equipment and machines
AI OCR
Automated data capture from images and labels
Label Printers
Direct label printing from Tulip apps
The Trusted Tulip Life Sciences Partner
- First Tulip Life Sciences partner
- Any size deployment - from single- site deployments up and Large global life sciences roll-outs, fully delivered and validated
- 30+ factories | 300+ validated apps
- Supporting multiple GxP use cases for leading global BioPharma companies
Tulip Services We Offer
Onboarding
Design, set up, and train teams to get Tulip live fast
Consulting & Advisory
Shape a clear Tulip digitalization, governance, and GxP strategy
Solution Delivery
Build scalable apps, data models, and integrations, including global standard apps.
Validation
Ensure compliance through a lean validation strategy and execution, tried and tested at 30+ sites
Support
Ongoing support to keep operations, COE’s, & Citizen Development running smoothly
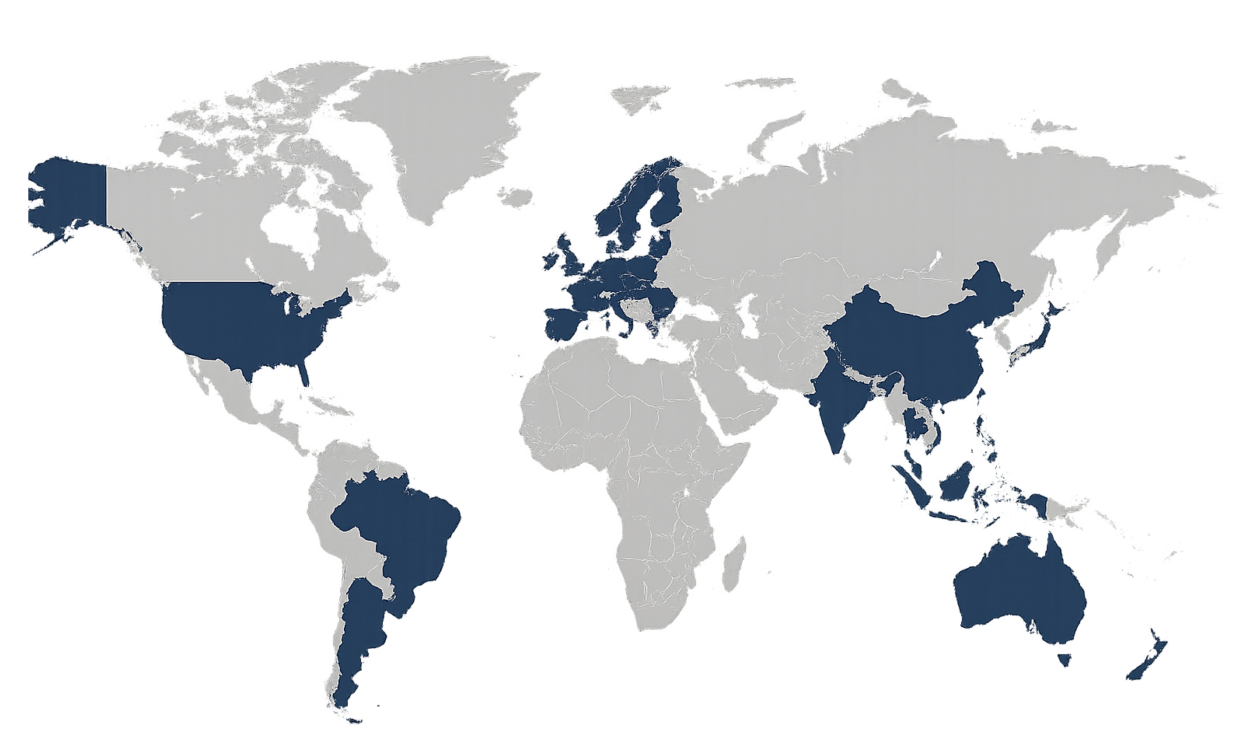
Proven Across Global Life Sciences Operations
Validated Tulip deployments delivered across Americas, Europe, Asia, and Africa for regulated manufacturers worldwide.

Factorytalk have distilled over 8 years of Tulip App development into a range of Tulip App templates available as products.
- Start with proven Tulip Apps from production hardened experience.
- Use as baselines for projects, adapted to you requirements, saving cost and time.
- Annual Support, maintenance, and version roadmaps available.
Tulip Lifecycle Manager (TLM)
End-to-end lifecycle and validation for all Tulip apps in GxP environments.
TLM brings governance, automated validation, and full traceability together
—so you can scale Tulip confidently from single sites to global operations.

“Factorytalk have been instrumental in our success deploying Tulip, acting as a trusted partner across the global programme, solution deployments, and validation. Their attitude, creativity, and high-quality services are a testament to the company.”

Bob Buhlmann
Head of Quality Digital and Computer Strategy, AstraZeneca
“What made the difference this time was the partnership. Factorytalk understood both the technology and the GMP reality of manufacturing. They didn’t push a big-bang MES — they helped us build something practical, scalable, and aligned with how our site actually works.”

David Li
IT Experience Lead, Consumer Health
Manufacturing Sites, Bayer


Bayer case study
See how Bayer Indonesia scaled smart manufacturing through a flexible, business-owned MES strategy— combining composable technology with GMP expertise.
The result
Faster releases, higher data integrity,
and a digital foundation that grows
with the business.
Want to see
what we can do for you?
See for yourself how Tulip can streamline your operations, improve collaboration and increase overall compliance.




