
David Margetts, Group Executive Director at Factorytalk, was honoured to speak at the Malaysian Organisation of Pharmaceutical Industries (MOPI) webinar on Artificial Intelligence in the Pharmaceutical Industry.
The session explored how AI is moving beyond experimentation and becoming operationally relevant in GxP-regulated pharmaceutical environments — and what this means for manufacturers across ASEAN.
Key takeaways from the session
The current state of AI in GxP manufacturing

AI adoption is accelerating globally, with organisations beginning to deploy AI agents across core manufacturing areas such as:
- Quality management
- Planning and logistics
- Supply chain optimisation
However, while AI adoption in Malaysia has increased significantly, many companies remain at basic efficiency levels rather than using AI to drive innovation.
A key question raised during the session was:
Can business leaders realistically implement, configure, maintain, and optimise AI systems while managing daily operations?
Where AI Is changing pharma operations today
Supply chain
- Improved GPS + RFID + IoT for real-time tracking
- AI route optimisation reducing fuel consumption and delivery times
- AI analysis of trade regulations and landed costs
Warehousing
- 20–35% faster picking speeds
- Storage density increased up to 4x
- 24/7 robotic operations
- Reduced error rates and returns
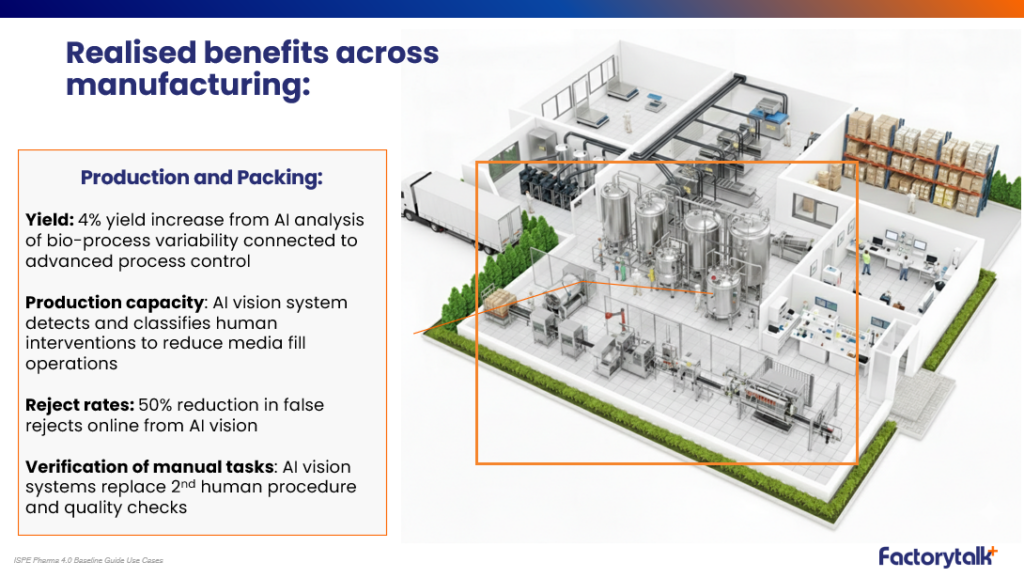
Production & packing
- 4% yield increase from AI-driven analysis of process variability
- 50% reduction in false rejects using AI vision systems
- Automated monitoring and classification of operator interventions
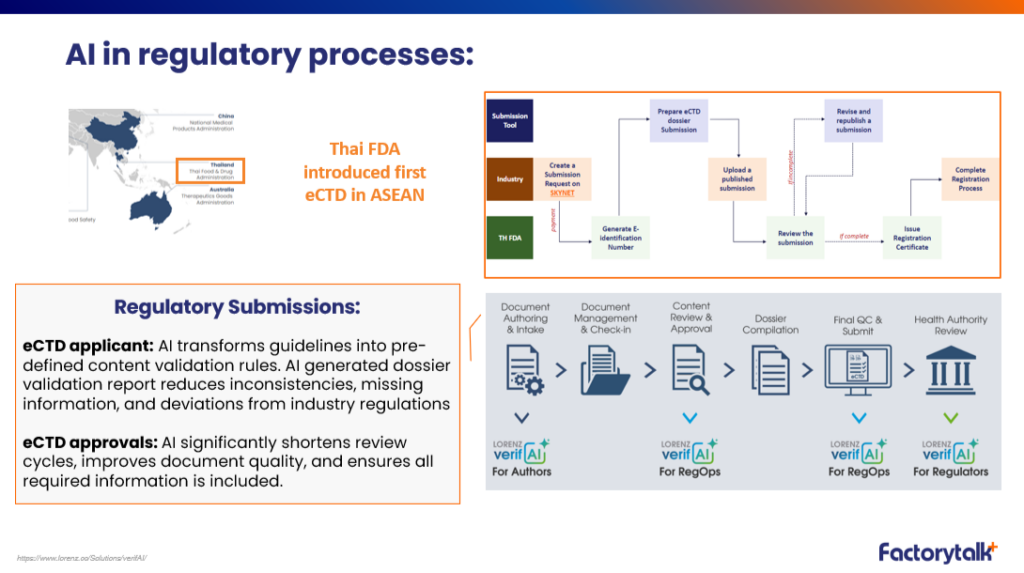
Regulatory submissions
- AI transforms regulatory guidelines into validation rules
- Reduced inconsistencies and missing information
- Faster review cycles and improved dossier quality
Regulatory evolution: Human-in-the-loop
Pharma regulators are progressively moving from traditional review processes toward automated verification models with human oversight.
Emerging guidance, including developments aligned with Annex 22 draft thinking, suggests that Human-in-the-Loop frameworks will be formalised as AI adoption expands in GxP environments.
Key considerations for AI in GxP
David highlighted that AI success depends not only on technology, but on governance:
- IP and data privacy protection
- Clear accountability and responsibility
- Data security and controlled network environments
- Model lifecycle management
- Explainability and trust in AI-generated outputs
Strong data governance and controlled integration are essential before scaling AI initiatives.
What this means for pharma
AI in pharmaceutical manufacturing is no longer theoretical — it is already delivering measurable benefits across supply chain, production, quality, and regulatory operations.
The real opportunity now is to implement AI responsibly, with structured validation, strong governance, and a clear human oversight model.
Factorytalk remains committed to supporting life sciences manufacturers in navigating this transition — turning AI from ambition into compliant, practical reality.
Want to learn more about AI in regulated manufacturing?
Get in touch with our team to discuss your digital readiness, governance framework, and practical next steps.




